A Country Doctor, But Oh So Much More: Francisco Lopera, 73 (Links to an external site)
On Tuesday September 10, Francisco Lopera died of cancer at his home in Medellin, Colombia. He was 73, and only just beginning to see his life’s work come to fruition.
The Knight Family DIAN-TU-003 Amyloid Removal Trial
MEMO DATE: 15 April 2024 TO: DIAN-TU-001 Gantenerumab Open Label Extension Participants FROM: Dr. Randall Bateman, Director, Knight Family DIAN-TU and Dr. Eric McDade, Associate Director, Knight Family DIAN-TU RE: DIAN-TU-003 Amyloid Removal Trial As previously announced by the DIAN-TU on 18 December 2023 and the Alzheimer’s Association on 09 April 2024, the Knight Family Dominantly Inherited Alzheimer […]
The Knight Family DIAN-TU Primary Prevention Trial Announcement
The statement below is an update to the 20 December 2022 announcement by the Knight Family Dominantly Inherited Alzheimer Network Trials Unit (DIAN-TU) regarding the DIAN-TU-002 Primary Prevention Trial. In late 2022, the Knight Family DIAN-TU paused the launch of the DIAN-TU-002 Primary Prevention Trial of gantenerumab due to discontinuation of the drug development program. […]
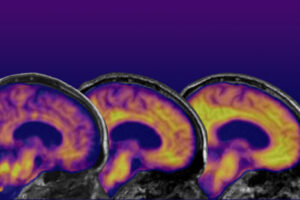
The New Age of Alzheimer’s (Links to an external site)
Moment of promise (Links to an external site)
Alzheimer’s Association and GHR Foundation Commit $4 Million to Extend Prevention Trial in Rare Alzheimer’s (Links to an external site)
The Knight Family DIAN-TU Amyloid Removal Trial
Update to the 18 August 2023 announcement by the Knight Family DIAN-TU)regarding the DIAN-TU-001 Open Label Extension (OLE) Study
Podcast: Newly approved drug may slow progression of Alzheimer’s (Links to an external site)
Lilly drug slows Alzheimer’s by 35%, bolstering treatment approach (Links to an external site)
Conquering Alzheimer’s: a look at the therapies of the future (Links to an external site)
Seeking Alzheimer’s clues from few who escape genetic fate (Links to an external site)
Lifetime Achievement Award (Links to an external site)
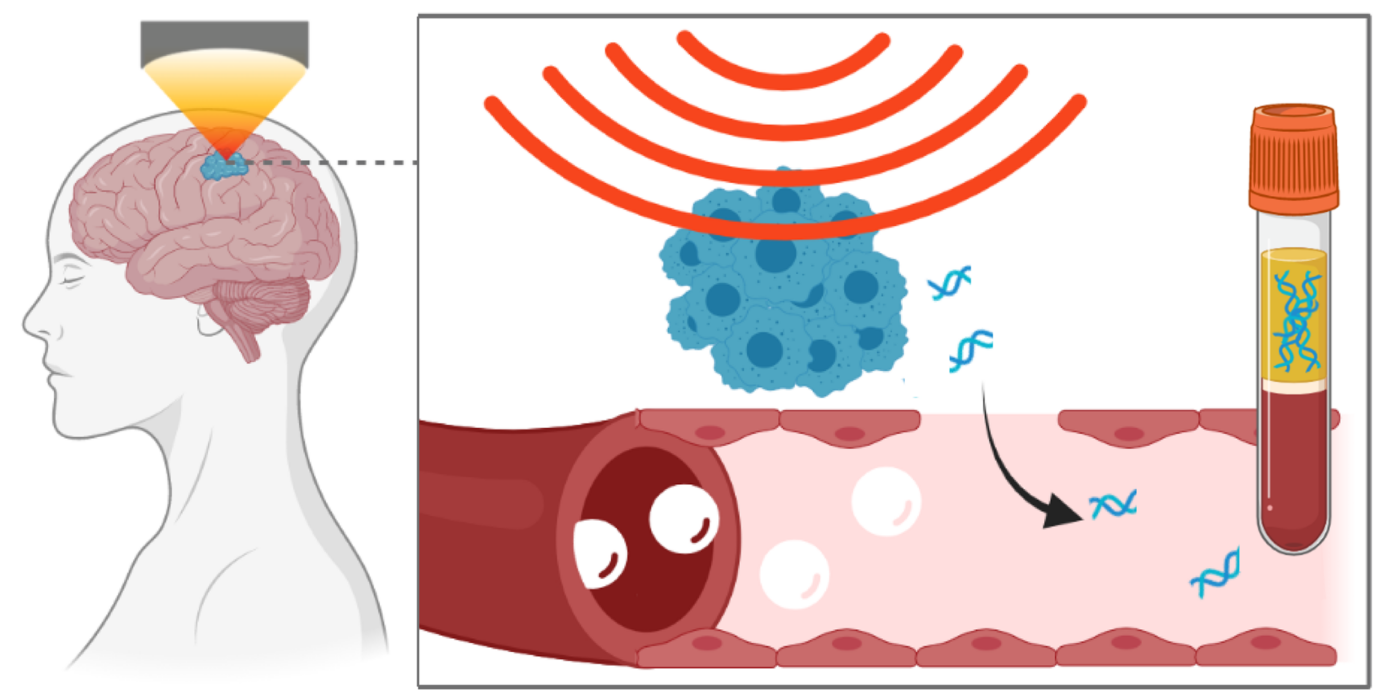
Focused ultrasound technique leads to release of neurodegenerative disorders biomarkers (Links to an external site)
Update on the DIAN-TU-001 Trial with E2814 and Lecanemab (Eisai Co., Ltd)
The statement below is in response to Eisai’s 06 January 2023 announcement regarding the FDA’s accelerated approval of lecanemab for the treatment of symptomatic Alzheimer’s disease (AD). On January 6th, 2023, the FDA announced approval of lecanemab (an anti-amyloid beta (Aβ) protofibril antibody) through its Accelerated Approval Program for the treatment of mild cognitive impairment […]
FDA APPROVES LEQEMBI™ (LECANEMAB-IRMB) UNDER THE ACCELERATED APPROVAL PATHWAY FOR THE TREATMENT OF ALZHEIMER’S DISEASE (Links to an external site)
Update on the DIAN-TU-002 Primary Prevention Trial
20 December 2022 Update on the DIAN-TU-002 Primary Prevention Trial The statement below is an update to the 15 November 2022 announcement by the Knight Family Dominantly Inherited Alzheimer Network Trials Unit (DIAN-TU) regarding the DIAN-TU-002 Primary Prevention Trial with Gantenerumab. During the presentation of topline results from GRADUATE I and II studies at the […]
WashU, Eisai form drug discovery collaboration (Links to an external site)
Update on the DIAN-TU-001 Open Label Extension and DIAN-TU-002 Primary Prevention Trials with Gantenerumab (Roche/Genentech)
The statement below is in response to the 14 November 2022 announcement regarding the results from the GRADUATE I and II AD studies found on Roche’s website. Roche, known as Genentech in the United States, and the Dominantly Inherited Alzheimer Network Trials Unit (DIAN-TU) at Washington University in St. Louis have been evaluating gantenerumab in […]
[Ad hoc announcement pursuant to Art. 53 LR] Roche provides update on Phase III GRADUATE programme evaluating gantenerumab in early Alzheimer’s disease (Links to an external site)
Update on the DIAN-TU-001 Trial with E2814 and Investigational Lecanemab (Eisai Co., Ltd)
The statement below is in response to the 28 September 2022 announcement found on Eisai’s website. Eisai Co., Ltd. (Headquarters: Tokyo, CEO: Haruo Naito, “Eisai”) and Biogen Inc. (Nasdaq: BIIB, Corporate headquarters: Cambridge, Massachusetts, CEO: Michel Vounatsos, “Biogen”) announced positive topline results from the large global Phase 3 confirmatory Clarity AD clinical trial of lecanemab […]
LECANEMAB CONFIRMATORY PHASE 3 CLARITY AD STUDY MET PRIMARY ENDPOINT, SHOWING HIGHLY STATISTICALLY SIGNIFICANT REDUCTION OF CLINICAL DECLINE IN LARGE GLOBAL CLINICAL STUDY OF 1,795 PARTICIPANTS WITH EARLY ALZHEIMER’S DISEASE (Links to an external site)
Roche provides update on Alzheimer’s Prevention Initiative study evaluating crenezumab in autosomal dominant Alzheimer’s disease (Links to an external site)
Could drugs prevent Alzheimer’s? These trials aim to find out (Links to an external site)
Blood test for Alzheimer’s highly accurate in large, international study (Links to an external site)
First Subject Enrolled in Phase II/III Study of Eisai’s Anti-MTBR Tau Antibody E2814 for Dominantly Inherited Alzheimer’s Disease (DIAD), conducted by DIAN-TU (Links to an external site)
New Alzheimer’s prevention trial in young people (Links to an external site)
$11.5 million commitment supports new Alzheimer’s prevention clinical trial (Links to an external site)
International Alzheimer’s clinical trial to test two drugs in combination (Links to an external site)
Global Incubator Seed Grants awarded (Links to an external site)
The Search for a Drug to End Alzheimer’s (Links to an external site)
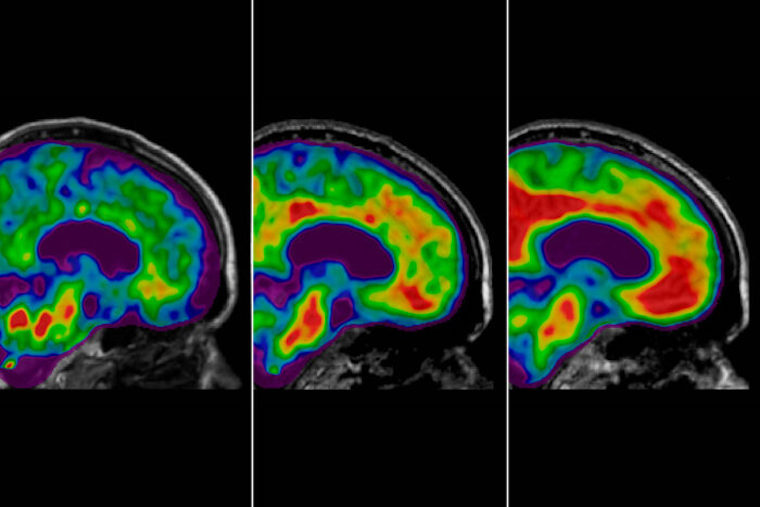
Some antiamyloids remove target plaques from the brain, yet don’t seem to halt cognitive decline. Researchers are working to understand why.